
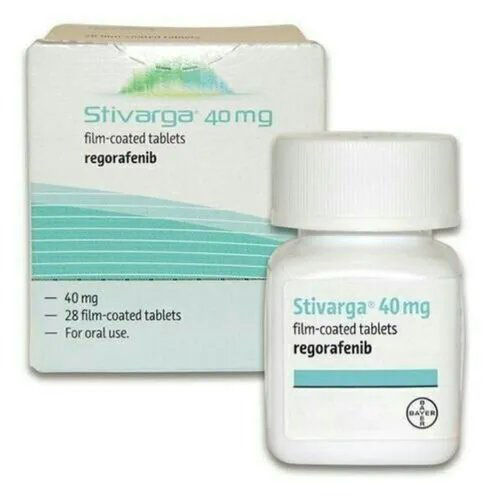
Stivarga Regorafenib 40 Mg Tablet
75000 INR/Bottle
Product Details:
- Dosage Form Tablet
- Indication Treatment of metastatic colorectal cancer, gastrointestinal stromal tumors (GIST), and hepatocellular carcinoma (HCC)
- Salt Composition Regorafenib 40 mg
- Enzyme Types Tyrosine Kinase Inhibitor (TKI)
- Feature Multi-kinase inhibitor with antiangiogenic and antiproliferative effects
- Ingredients Regorafenib; Excipients
- Application Oral administration for oncology therapy
- Click to View more
X
Stivarga Regorafenib 40 Mg Tablet Price And Quantity
- 1 Bottle
- 75000 INR/Bottle
Stivarga Regorafenib 40 Mg Tablet Product Specifications
- Light brown, round, film-coated tablets
- Tyrosine Kinase Inhibitor (TKI)
- Multi-kinase inhibitor with antiangiogenic and antiproliferative effects
- Treatment of metastatic colorectal cancer, gastrointestinal stromal tumors (GIST), and hepatocellular carcinoma (HCC)
- Odorless
- Regorafenib; Excipients
- Store below 30C in a dry place, protect from light and moisture
- Regorafenib 40 mg
- Oral administration for oncology therapy
- 24-36 months (refer to packaging)
- Tablet
- Advise on potential side effects, liver monitoring required
- L01XE21
- Oral
- Known hypersensitivity to Regorafenib or any excipients
- US FDA and other regulatory authorities (refer to local approvals)
- Stivarga
- Yes, prescription only
- 28 tablets per box/pack
- Film-coated tablet
Product Description
Product details
- Country of Origin : Made in India
- Shelf life : 36 month
- Form : Tablet
- Packaging Size : 28 Tablets
- Prescription/Non prescription : Non prescription
- Treatment : Anti cancer
- Composition : Regorafenib
- Brand : Stivarga
Comprehensive Cancer Therapy
Stivarga Regorafenib 40 mg tablet is specifically formulated as an oral treatment for advanced stages of colorectal cancer, GIST, and hepatocellular carcinoma. Its targeted mechanism as a tyrosine kinase inhibitor disrupts cancer cell growth and spread, offering advanced support for oncology patients seeking improved treatment outcomes, under appropriate medical guidance.
Key Features and Safety
Each film-coated tablet contains 40 mg of Regorafenib, provided in odorless, light brown, round tablets. The formulation is designed for oral use and should be consumed with guidance from a healthcare professional. Patient counselling focuses on monitoring potential side effects and liver function, with contraindications for known hypersensitivity to Regorafenib or its components.
Storage and Shelf Life
Stivarga tablets should be stored in a dry location below 30C, protected from light and moisture to maintain potency. The shelf life ranges between 24 to 36 months-always check packaging for specific expiry dates. These instructions facilitate proper storage and help prevent degradation of the medication.
FAQ's of Stivarga Regorafenib 40 Mg Tablet:
Q: How should Stivarga Regorafenib 40 mg tablets be taken?
A: Stivarga tablets are administered orally, usually once daily, as prescribed by your oncologist. Always follow the exact dosing schedule and recommendations given by your healthcare provider.Q: What conditions is Stivarga indicated for?
A: Stivarga is indicated for the treatment of metastatic colorectal cancer, gastrointestinal stromal tumors (GIST), and hepatocellular carcinoma (HCC). It is intended for patients with advanced disease who meet the criteria specified by their physician.Q: When is patient counselling recommended during Stivarga therapy?
A: Patient counselling should occur at the start of therapy and throughout treatment to discuss potential side effects, the necessity of liver monitoring, adherence to dosage, and to recognize symptoms of hypersensitivity.Q: Where should Stivarga tablets be stored for optimal preservation?
A: The tablets should be stored in a dry place, below 30C, and away from light and moisture. This ensures the medication retains its effectiveness throughout its shelf life.Q: What process should be followed for monitoring during Stivarga treatment?
A: Regular liver function tests and general health assessments are necessary, as advised by your healthcare provider, to track potential side effects and efficacy during therapy.Q: What are the benefits of using Stivarga Regorafenib in cancer therapy?
A: Stivarga functions as a multi-kinase inhibitor with antiangiogenic and antiproliferative effects, slowing tumor growth and spread. It offers therapeutic value in managing advanced cancers, with a proven track record in clinical studies.Q: Who should avoid taking Stivarga Regorafenib tablets?
A: Individuals with a known hypersensitivity to Regorafenib or any tablet excipients should not take this medication. Always inform your healthcare provider about allergies and medical history before beginning treatment.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email









 Call Me Free
Call Me Free
