
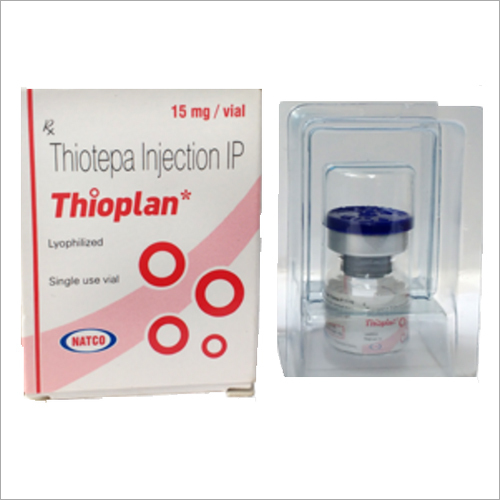
Thioplan 15 MGVial (Thiotepa)
1200 INR/Piece
Product Details:
- Dosage Form Lyophilized Powder for Injection
- Salt Composition Thiotepa 15 mg/vial
- Indication Indicated for the treatment of certain cancers, including ovarian, breast, and bladder cancer.
- Enzyme Types Not applicable (Cytotoxic agent, not enzyme-based)
- Feature Other
- Ingredients Other
- Application Other
- Click to View more
X
Thioplan 15 MGVial (Thiotepa) Price And Quantity
- 1200 INR/Piece
- 100 Piece
- Intravenous
- Celon Laboratories Ltd.
- Reconstitute with sterile water for injection before administration
- Approved by Drug Regulatory Authorities as per local requirements
- Antineoplastic (Chemotherapy) Agent
- Celon Laboratories Ltd.
- To be handled by qualified healthcare professionals only; cytotoxic product
- Prescription only (Rx)
- Vial
- 1 vial per pack
Thioplan 15 MGVial (Thiotepa) Product Specifications
- Other
- Cream Powder
- Thiotepa 15 mg/vial
- Cold & Dry
- Not applicable (Cytotoxic agent, not enzyme-based)
- Other
- Other
- 2 years from the date of manufacture
- Indicated for the treatment of certain cancers, including ovarian, breast, and bladder cancer.
- Lyophilized Powder for Injection
- Intravenous
- Celon Laboratories Ltd.
- Reconstitute with sterile water for injection before administration
- Approved by Drug Regulatory Authorities as per local requirements
- Antineoplastic (Chemotherapy) Agent
- Celon Laboratories Ltd.
- To be handled by qualified healthcare professionals only; cytotoxic product
- Prescription only (Rx)
- Vial
- 1 vial per pack
Thioplan 15 MGVial (Thiotepa) Trade Information
- 1000 Piece Per Day
- 3 Days
Product Description
Thioplan 15 MGVial (Thiotepa), manufactured and marketed by Celon Laboratories Ltd., is an elite antineoplastic chemotherapy agent highly recommended for oncology specialists. It contains a sterling 15 mg/vial Thiotepa, presented as a lyophilized cream powder, and must be reconstituted with sterile water before intravenous administration. Exceptional for treating certain cancers, including ovarian, breast, and bladder cancer, it is prescription-only and must be handled by qualified professionals due to its cytotoxicity. With a two-year shelf life and approved by local drug regulatory authorities, it assures outstanding efficacy and safety. Exported from India by certified suppliers, the product offers reliable quality with each vial carefully packed for optimal storage and handling.
Competitive Advantages and Outstanding Features
Thioplan 15 MGVial (Thiotepa) offers exceptional efficacy in cancer therapy, making it highly trusted by healthcare professionals worldwide. Its competitive advantage lies in its broad application across ovarian, breast, and bladder cancers, suitable for various treatment protocols. The cream powder formulation ensures optimal reconstitution and stability, while strict manufacturing standards guarantee a reliable product. Its outstanding shelf life, as well as precise dosage consistency, affirms Thioplan as an elite choice in antineoplastic care.
Sample Policy, Packing & Dispatch Details
Sample requests for Thioplan 15 MGVial (Thiotepa) are welcomed, with reasonable outlay arrangements depending on policy. Each product is individually packed using robust, tamper-evident packaging to maintain safety during goods transport. Packing & dispatch procedures are managed expertly to ensure prompt and secure delivery. All vials are dispatched in accordance with regulatory guidelines, ensuring sample availability to qualified buyers for evaluation, trading, or export across India and to international clients.
Competitive Advantages and Outstanding Features
Thioplan 15 MGVial (Thiotepa) offers exceptional efficacy in cancer therapy, making it highly trusted by healthcare professionals worldwide. Its competitive advantage lies in its broad application across ovarian, breast, and bladder cancers, suitable for various treatment protocols. The cream powder formulation ensures optimal reconstitution and stability, while strict manufacturing standards guarantee a reliable product. Its outstanding shelf life, as well as precise dosage consistency, affirms Thioplan as an elite choice in antineoplastic care.
Sample Policy, Packing & Dispatch Details
Sample requests for Thioplan 15 MGVial (Thiotepa) are welcomed, with reasonable outlay arrangements depending on policy. Each product is individually packed using robust, tamper-evident packaging to maintain safety during goods transport. Packing & dispatch procedures are managed expertly to ensure prompt and secure delivery. All vials are dispatched in accordance with regulatory guidelines, ensuring sample availability to qualified buyers for evaluation, trading, or export across India and to international clients.
FAQ's of Thioplan 15 MGVial (Thiotepa):
Q: How should Thioplan 15 MGVial (Thiotepa) be reconstituted before administration?
A: Reconstitute Thioplan 15 MGVial (Thiotepa) with sterile water for injection as directed by healthcare protocols prior to intravenous use. Only qualified healthcare professionals should undertake this process, following cytotoxic handling guidelines.Q: What is the main benefit of using Thioplan 15 MGVial (Thiotepa) for cancer treatment?
A: Thioplan 15 MGVial (Thiotepa) provides exceptional efficacy in the management of ovarian, breast, and bladder cancers, offering elite therapeutic outcomes while being supported by stringent quality standards.Q: When should Thioplan 15 MGVial (Thiotepa) be administered?
A: Thioplan 15 MGVial (Thiotepa) must be administered only under medical supervision and as prescribed by an oncologist, typically as part of a planned chemotherapy regimen for indicated cancers.Q: Where should the vials be stored after purchase?
A: Vials should be stored in a cold and dry environment as per label instructions to preserve potency and extend shelf life, which is two years from the date of manufacture.Q: What application surfaces is Thioplan 15 MGVial (Thiotepa) suitable for?
A: This product is intended for intravenous administration in a clinical setting, suitable only for application by trained medical professionals in hospitals or specialized cancer centers.Q: How can I obtain a sample of Thioplan 15 MGVial (Thiotepa)?
A: Samples are available to qualified buyers and institutions. Interested parties should contact the supplier, exporter, or manufacturer for sample policy details and arrange outlay and dispatch as needed.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email









 Call Me Free
Call Me Free
